|
12/27/2023 0 Comments Oxygen atom
Build double or triple bonds depending upon the number of electrons the central atom has.Ĭonclusion Construction of NO3 Lewis Dot Structureġ. The central atom must complete its octet. 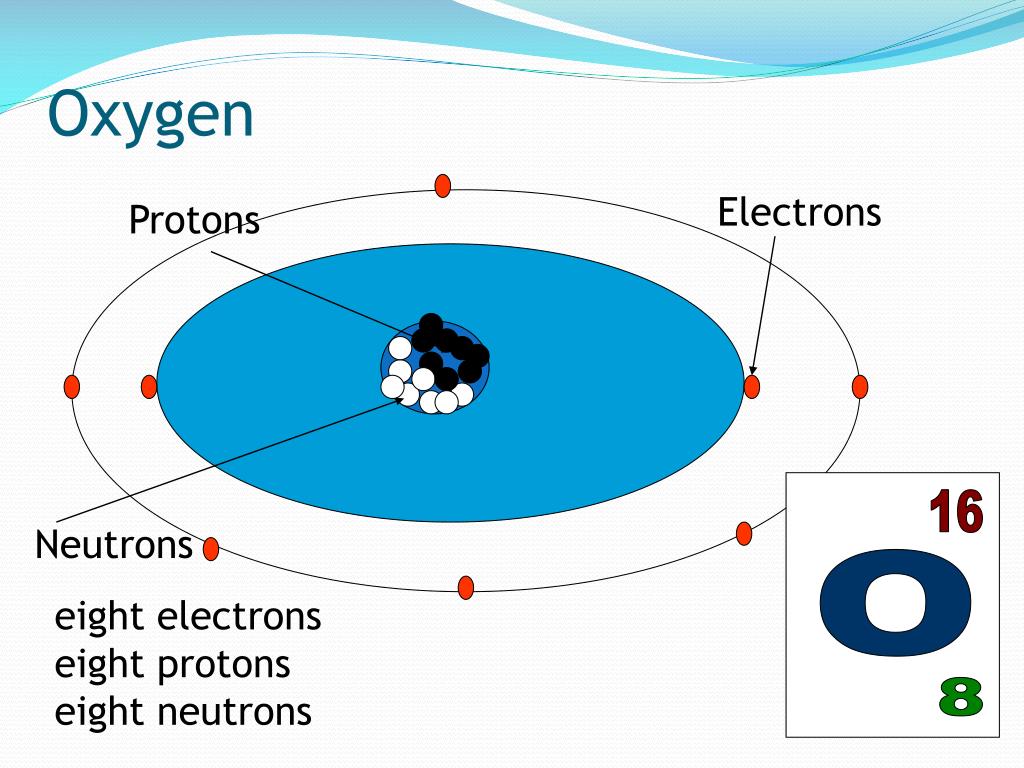
Assign the lone pairs to the terminal atoms and make sure each completes its last shell with 8 electrons.Ĩ. 
Lone pairs electrons = Valence electrons – Bonding electronsħ. Calculate the lone pairs electron using the below formula. Make a single bond and count the number of bonding electrons.Ħ. After deciding the central atom, arrange the most electronegative atoms surrounding it.ĥ. (Bonding pairs involve the number of electrons that take part in bonding between atoms and lone pairs have electrons that do not help atoms in bonding.)Ĥ. Then find out the number of bonding pairs and lone pairs. Depending upon the number of atoms in a compound, calculate the total number of valence electrons.ģ. (Exceptions are hydrogen and boron elements)Ģ. Follow the octet rule where an atom should complete its outermost shell by the total number of 8 electrons. Lewis introduced the concept of electron dot structure.īelow are some rules to frame any compound’s Lewis dot structure.ġ. NO3 is easily soluble water but too much concentration in drinking water is harmful to human health that affects blood carrying oxygen. It also treats heart pains.īoth nitrogen and oxygen are important to an ecosystem that includes flora and fauna. It is used as fertilizers (like ammonium, sodium, potassium) in agricultural farms for higher solubility and biodegradability. Nitrate is an important source of nitrogen and oxygen. The compound has its chemical name as nitrate formed after nitric acid looses a proton from it. So, it is also referred to by the name of nitrogen oxoanion. Select the oxygen and click the "Decrease formal charge" button to adjust the formal charge to -1 or -2 if you want a hydroxyl or oxygen ion ligand, respectively.NO3 is a polyatomic ion with a negative charge.To adjust the iron-oxygen distance, right click one of them while both are selected, choose "Adjust Distance" and then adjust the distance appropriately.Then click "Decrement bond order" to change it to a zero-order bond. Select the oxygen and the iron, and in the BUILD tool click the "Add bond between two selected atoms" button.Click above the iron atom where you want to place the oxygen.In the BUILD tool, choose "Draw", and choose O for the element in the Draw Structure banner. 
(Use Shift with the middle mouse button to ensure rotation about y only, and drag horizontally.) Rotate the structure around the y axis so you can see the iron atom.They should be aligned vertically in the Workspace, with the sulfur below the iron. Click the CYS sulfur, then the iron atom.Select Y for aligning two atoms to an axis, and select "Pick atoms" underneath this option.Choose View → Align to open the Align Atoms panel.Use the "Fit to Selection" button in the main toolbar to zoom in on the heme group, if you didn't use the Find tool.Select the heme group in the STRUCTURE HIERARCHY tree under its protein chain, or use the Find tool (Select → Find) to select the heme.You can use the following instructions to add the oxygen atom. It is helpful to align the heme unit first, so that it's easy to draw in the right direction. The best way of adding an oxygen atom is to use the Draw Structure feature on the Build toolbar, for which valence violations are ignored.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
